
You should deactivate any pop-up blockers, spam filters, and company firewalls that could prevent the WebEx client platform or web-based course from working properly. If successful, you will see a screen that says "Congratulations! Your system is now set up properly. To join a test session (meeting), click here and enter your name and email address to join. If you do not have administrative privileges on the SAME computer you will be using the day of your training (i.e., your workstation within your organization) you may need the assistance of your local IT/IS help desk staff for the platform to engage correctly. This can help prevent problems launching content the day your training begins.
We strongly suggest that you join a WebEx test session in advance of your scheduled virtual training, using your preferred web browser. WebEx – Virtual Course: Advance Preparation and Student Information Join a Test Session
#ISO 17025 2017 ANDROID#
Mobile: Safari in Apple iOS 10 or later, Google Chrome in Apple iOS 10 or later, Google Chrome in Android OS 4.4 or later ISO/IEC 17025:2017 specifies the general requirements for the competence, impartiality and consistent operation of laboratories.Mac: Safari (latest version), Google Chrome (latest version), Firefox (latest version).Windows: Internet Explorer 11, Microsoft Edge (latest version), Google Chrome (latest version), Firefox (latest version) 17025:2017 standard covers the newest ISO/IEC terminology and the changes that have been included in the International Vocabulary of.This includes mobile devices such as iPhones, iPads, and Android-enabled phones and tablets. You can view courses on any devices that support these browsers. Technical Requirements (impacts all ASQ-owned eLearning) For virtual WebEx courses, you will receive another email 24 hours prior to the start of class containing additional access information. If the email is not there, please contact ASQ. If you do not, please check your email junk, spam, or clutter folders. Here is what you need to participate in an ASQ web-based, self-paced, or instructor-led virtual WebEx course.Īfter course registration, you will receive an email with access instructions. Management System RequirementsĨ.2 Management System Documentation (Option A)Ĩ.3 Control of Management System Documents (Option A)Ĩ.5 Actions to Address Risks and Opportunities (Option A)Īdditional Requirements of Accreditation Bodies (AB’s) System Requirements This accreditation is intended for all organizations performing tests and/or calibration, including first-, second- and third-party laboratories and laboratories where testing and/or calibration forms part of inspection and product certification.6.3 Facilities and Environmental ConditionsĦ.6 Externally Provided Products and Servicesħ.1 Review of Requests, Tenders, and Contractsħ.2 Selection, Verification, and Validation of Methodsħ.4 Handling of Test or Calibration Itemsħ.6 Evaluation of Measurement Uncertaintyħ.11 Control of Data and Information Management Other changes include revised terminology as well as an updated scope that now covers all laboratory activities including the testing, calibration, and sampling that is associated with subsequent calibration and testing. It incorporates the use of computer systems and electronics as well as focusing on the results of the process over the task. The most popular standard for the competence of testing and.
#ISO 17025 2017 ISO#
This revision places a stronger emphasis on information technologies due to its increased impact on the industry. ISO 17025:2017 is an International Quality Standard for Testing and Calibration Laboratories.
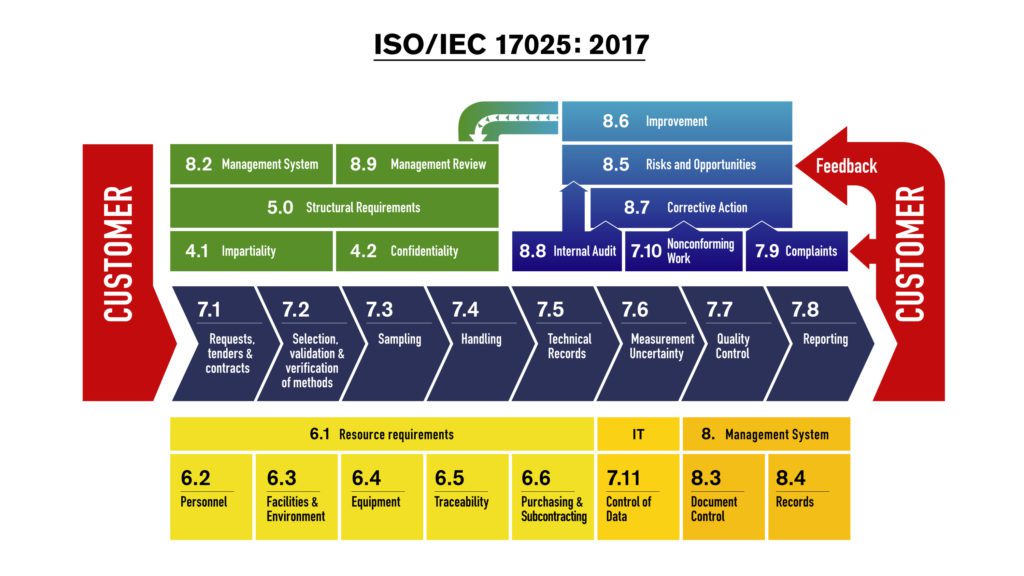
The standard outlines requirements for quality management in a laboratory's specific testing areas. Laboratory customers, regulatory authorities, organizations and schemes using peer-assessment.

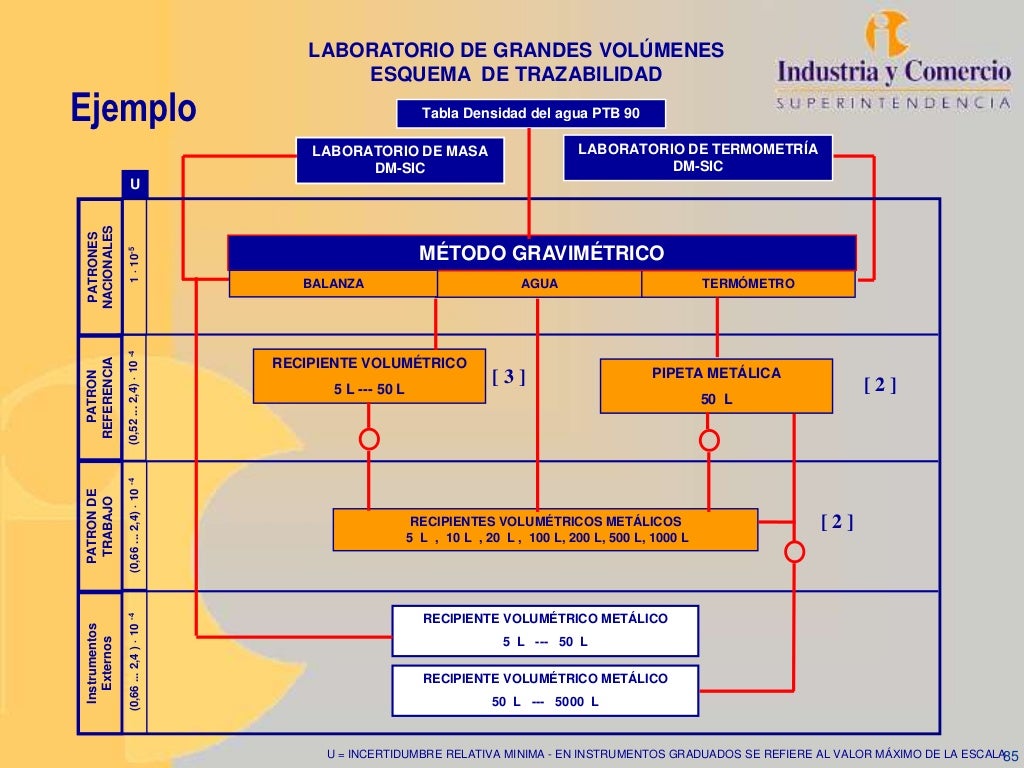
ISO/IEC 17025:2015 is a company-level accreditation based on a standard published by the International Organization for Standardization (ISO) titled "General requirements for the competence of testing and calibration laboratories." ISO/IEC 17025:2017 specifies the general requirements for the competence, impartiality and consistent operation of laboratories.
